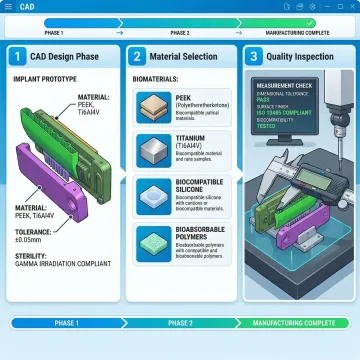
What is medical device prototyping?
Medical device prototyping is the process of creating physical models of medical devices to validate design, functionality, and manufacturability before committing to full-scale production. It involves rapid fabrication of prototype components using advanced manufacturing techniques to test structural integrity, ergonomics, biocompatibility, and regulatory compliance. At Evok, we offer comprehensive prototyping services including CAD design review, material selection for biocompatibility, prototype fabrication, analytical testing, and first article qualification with complete documentation for FDA submissions.
How much does it cost to have a prototype made?
Medical device prototype costs typically range from $100-$500 per prototype depending on size, complexity, material requirements, and the prototyping system used. Our pricing includes engineering review, design modifications for moldability, material selection guidance, and prototype fabrication. We're very upfront with costs so you can make informed decisions about your development budget. Contact us with your specific requirements for a detailed quote that accounts for your device's unique specifications and regulatory requirements.
What is FDA compliant mean?
FDA compliant means your medical device prototype meets the regulatory requirements set by the U.S. Food and Drug Administration for safety, efficacy, and quality. This includes using biocompatible materials, following Good Manufacturing Practices (GMP), maintaining detailed documentation, and meeting specific performance standards for your device classification. At Evok, we ensure FDA compliance through rigorous material selection, comprehensive testing protocols, detailed first article qualification with 30 inspection points per cavity for critical dimensions, and complete documentation packages that support your regulatory submissions and design validation process.
What prototyping methods do you offer for medical devices?
We offer multiple rapid prototyping methods tailored to your validation needs, including injection molding prototypes, 3D printed functional models, and machined components. Our team helps you select the optimal prototyping system based on whether you need cosmetic verification or functional testing. We consider factors like material properties, surface finish requirements, mechanical performance, and sterilization compatibility to recommend the best approach for your medical device prototype.
How long does medical device prototype development take?
Timeline varies based on design complexity and testing requirements, but our streamlined process typically delivers prototypes in 2-4 weeks from design approval. This includes CAD review and modifications, material selection, prototype fabrication, and initial testing. First article qualification with complete documentation adds 1-2 additional weeks. Our speed-to-market focus and cross-functional team approach with continuous improvement mindset helps accelerate your development timeline without compromising quality or regulatory compliance.
Can you help with material selection for biocompatibility?
Absolutely. Material selection is critical for medical device prototypes, and our expertise in material and texture science ensures your device meets biocompatibility requirements. We evaluate materials based on ISO 10993 standards, sterilization method compatibility (autoclave, EtO, gamma radiation), chemical resistance, mechanical properties, and long-term performance. Our team guides you through FDA-approved material options that optimize both prototype performance and cost-effectiveness while meeting regulatory requirements.
What documentation do you provide for regulatory submissions?
We provide comprehensive FDA-ready documentation including detailed molding trial reports, first article qualification data with 30 inspection points per cavity for critical dimensions, process parameter monitoring (fill speed, temperatures, cycle times), Cp and Cpk statistical data, material certifications, and tolerance analysis. This complete documentation package supports your Design History File (DHF), design validation, and regulatory submissions for 510(k) or PMA approval processes.
Do you offer design modifications for manufacturability?
Yes, our CAD design services include expert engineering review and modifications to optimize your medical device for injection molding manufacturability. We implement changes for moldability, adjust wall thickness, optimize gate and vent sizing, and provide detailed redlining documentation of all modifications. Our goal is to ensure your prototype accurately represents what can be achieved in production while maintaining design intent, functionality, and regulatory compliance. This upfront design optimization saves significant time and cost in the long run.